Ingredients --
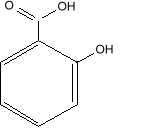
Salicylic Acid
Chemical Formula: C6H4(OH)CO2H
Synonyms
2-hydroxybenzoic acidDescription
White odorless crystalsUses
Salicylic acid is used to treat acne, warts, dandruff, psoriasis, and similar conditions. In the treatment of acne, it slows the shedding of skin cells in hair follicles, so they do not clog the pores and cause pimples. It also has a keratolytic effect, causing dead cells to slough off. The top layer of skin is removed, and pores unclogged. In treatments for warts, a stronger solution is used. This not only softens the wart so it can be rubbed off, but the irritation stimulates the immune system to attack the human papillomavirus that causes warts. Salicylic acid reacts with acetic acid to produce acetylsalicylic acid the active ingredient in aspirin. It reacts with methanol to form methyl salicilate, more commonly known as oil of wintergreen. Salicylic acid is common in many plants, such as broccoli, peppers, curry, cucumbers, and raisins, among many others. Salicylic acid reduces inflammation, and can reduce it in arteries to prevent hardening and narrowing.salicylic acid: InChI=1/C7H6O3/c8-6-4-2-1-3-5(6)7(9)10/h1-4,8H,(H,9,10)/f/h9H
methanol: InChI=1/CH4O/c1-2/h2H,1H3
By Simon Quellen Field