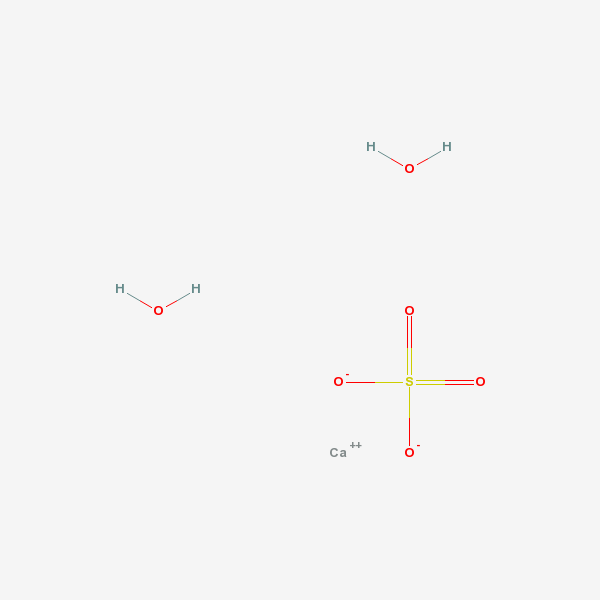
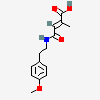
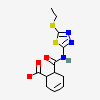
gypsum
 PubChem Notes:
PubChem Notes:
Calcium Sulfate A calcium salt that is used for a variety of purposes including: building materials, as a desiccant, in dentistry as an impression material, cast, or die, and in medicine for immobilizing casts and as a tablet excipient. It exists in various forms and states of hydration. Plaster of Paris is a mixture of powdered and heat-treated gypsum.
Gypsum - definition from Biology-Online.org
[(Science: chemical) a mineral consisting of the hydrous sulphate of lime (calcium). When calcined, it forms plaster of paris. selenite is a transparent, crystalline variety; alabaster, a fine, white, massive variety. Origin: L. gypsum, gr.; cf. ar. jibs plaster, mortar, Per. Jabsn lime. Its chemical formula is CaSO4•2H2O.
MediLexicon gypsum - Medical Dictionary Definition for Term 'gypsum'
[1. The natural hydrated form of calcium sulfate; a component of the stones, plasters, and investments used in dentistry.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|






















