Ingredients --
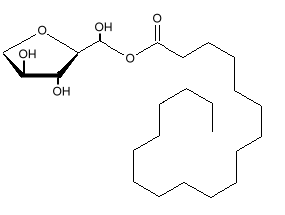
Sorbitan monostearate
Chemical Formula:
Sorbitan monostearate
Synonyms
SPAN 60Description
Wax-like creamy white powder. Sorbitan monostearate is an ester of sorbitol and stearic acid. The sorbitol end of the molecule is very soluble in water. The stearic acid end is soluble in fats. These properties make the molecule very good at making emulsions of oil and water. It is sometimes known as a synthetic wax.Uses
Sorbitan monostearate is used as an emulsifying agent in cake mixes, icings, baked goods, puddings, imitation whipped cream, hemorrhoid creams, and creams to treat dry skin. It is often used with polysorbate 80. Similar molecules can be made using other fatty acids, such as the shorter chained lauric acid.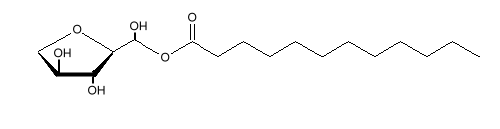
Sorbitan monolaurate
Chemistry Lesson
An emulsion is a mixture of oil and water. Some emulsions like butter and margarine, have tiny droplets of water in the oil. Others, like cream or mayonnaise, are droplets of oil in water. The choice of emulsifier has a large part to play in which kind of emulsion you get. Emulsifiers that are soluble in water generally have shorter chains of fats. These make emulsions of oil in water. Emulsifiers that have long chain fats, or several chains, are more soluble in oil than they are in water. These make emulsions of water in oil. The emulsifier polysorbate 80 is at the water-soluble end of the spectrum, making good emulsions of oil in water. The emulsifier glycerol monostearate is closer to the oil soluble end of the spectrum, making good emulsions of water in oil. Somewhere in the middle are soy lecithin and sorbitan monolaurate, which make good foam stabilizers in whipped cream and similar products.sorbitan monostearate: InChI=1/C24H46O6/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-22(27)29-19-21(26)24-23(28)20(25)18-30-24/h20-21,23-26,28H,2-19H2,1H3/t20-,21?,23+,24-/m1/s1
polysorbate 80: InChI=1/C32H60O10/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-30(36)40-25-24-37-26-28(38-21-18-33)32-31(41-23-20-35)29(27-42-32)39-22-19-34/h9-10,28-29,31-35H,2-8,11-27H2,1H3/b10-9+
stearic acid: InChI=1/C18H36O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20/h2-17H2,1H3,(H,19,20)/f/h19H
By Simon Quellen Field