Ingredients --
Benzoyl peroxide
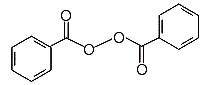
Chemical Formula: (C6H5CO)2O2
Synonyms
Description
White or off-white needle shaped water-soluble crystalsUses
Benzoyl peroxide is used as a bleaching agent in wheat flour, but its more familiar use is as an acne medication. Benzoyl peroxide has several benefits as an acne medication. It causes dead skin cells to slough off, like salicylic acid and resorcinol. It kills the bacterium Porpionibacterium acnes that causes acne pimples. It has an anti-inflammatory effect that reduces pimples and the pain they cause. It is a bleaching agent, oxidizing dyes to make them colorless. It eliminates fatty acids on the skin.Chemistry lesson
A peroxide is a compound that has an oxygen-oxygen bond. That is, it has two oxygen atoms bound together. Look at the structural formula at the top of the page. The molecule is symmetrical, with two identical parts joined at their oxygen atoms with an oxygen-oxygen bond. A familiar peroxide is hydrogen peroxide. It has two oxygen atoms bound together, and each of those atoms in turn it bound to a hydrogen atom. Peroxides make good bleaches. The oxygen atoms combine with dyes to make the dyes colorless. This process (called oxidation) can kill germs.benzoyl peroxide: InChI=1/C14H10O4/c15-13(11-7-3-1-4-8-11)17-18-14(16)12-9-5-2-6-10-12/h1-10H
resorcinol: InChI=1/C6H6O2/c7-5-2-1-3-6(8)4-5/h1-4,7-8H
hydrogen peroxide: InChI=1/H2O2/c1-2/h1-2H
salicylic acid: InChI=1/C7H6O3/c8-6-4-2-1-3-5(6)7(9)10/h1-4,8H,(H,9,10)/f/h9H
By Simon Quellen Field