Ingredients --
Hydrogen peroxide
Chemical Formula: H2O2
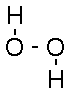
Synonyms
Description
Clear colorless liquid.Uses
Hydrogen peroxide is used as a topical antiseptic in dilute solutions, and as a water purifier in stronger solutions. It is also famous as a hair bleach. Hydrogen peroxide breaks down into water and oxygen. An liter of 3% hydrogen peroxide will generate 10 liters of oxygen when a catalyst is used to facilitate the breakdown. Catalysts can be metals such as iron, copper, or silver, or organics such as the enzyme catalase found in blood (it contains a heme group, like hemoglobin does, and the iron catalyzes the reaction). Catalase is an important enzyme in cells because hydrogen peroxide is a byproduct of metabolism, and can poison the cell unless it is decomposed quickly. Hydrogen peroxide is also produced by cells in the immune system, and catalase removes the excess. The 3% hydrogen peroxide you get at the drugstore is often protected from decomposing by the addition of sodium silicate, magnesium sulfate, or tin compounds. These stabilizers lock up the iron, copper, and other transition metals that can act as catalysts.hydrogen peroxide: InChI=1/H2O2/c1-2/h1-2H
By Simon Quellen Field